














| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |
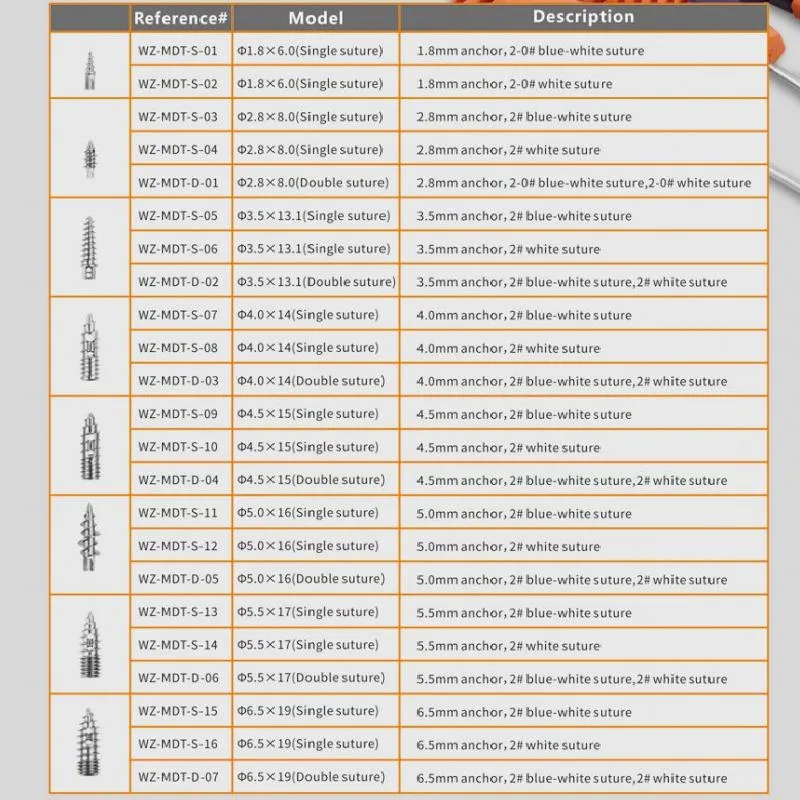
Titanium Alloy 5 mm Suture Anchor for Joint Arthroscopy System
The Suture Anchor System consists of an anchor, nonabsorbable suture, and inserter. The anchor is manufactured from Ti6Al4V alloy, strictly adhering to ISO5832-3:2016 standards. The suture is crafted from ultra-high molecular weight polyethylene (UHMWPE) material, woven without coating for optimal performance, meeting ASTM F2848-17 requirements.
The system features blue sutures dyed with FDA-approved materials for high visibility during surgery. The inserter parts that contact the human body are made of high-grade stainless steel (ASTM F899-12b), while the handle is ergonomically designed using polycarbonate and ABS materials.




Products are secured in professional blister boxes combined with protective outer cartons to ensure sterility and safety during transit.


 Tonk Orthopedic Implants
Tonk Orthopedic Implants