




1 / 5










| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |
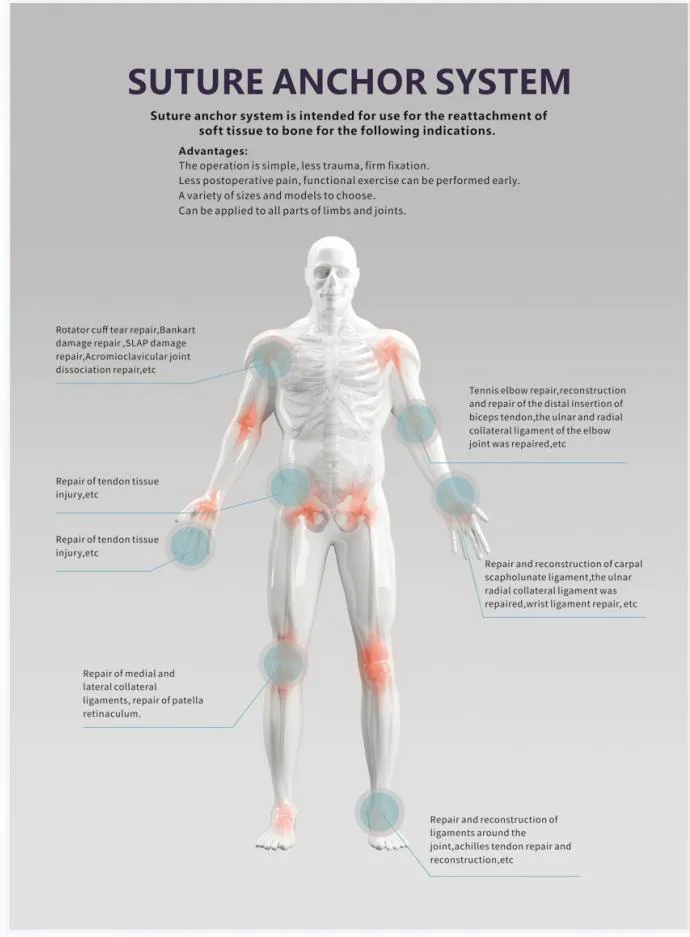
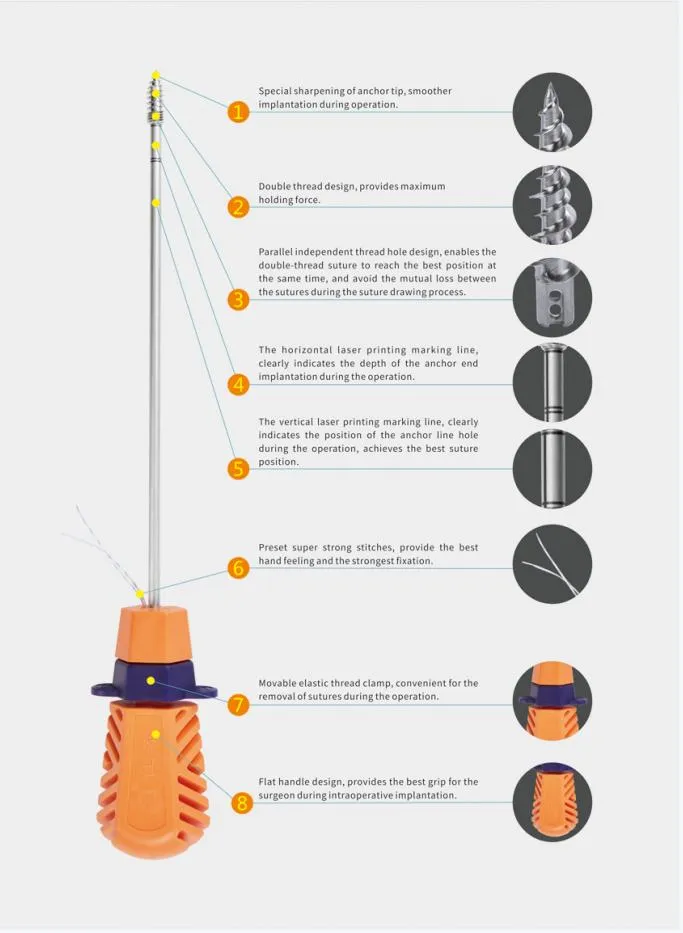
Suture Anchor System consists of anchor, nonabsorbable suture and inserter. Anchor is made of Ti6Al4V alloy, which meets the requirement of ISO5832-3:2016. Suture is made of ultra high molecular weight polyethylene material without coating, it is woven from several UHMWPE yarn, which meets the requirement of ASTM F2848-17. Blue suture is from white ultra high molecular weight polyethylene, dyed with blue color material, code of blue material is 73.1015, which listed and approved by FDA. Part of inserter contacting human body is made of stainless steel conforming ASTM F899-12b. Handle of inserter will not contact human body, and it is made of polycarbonate and ABS material.








| Item | Value |
|---|---|
| Properties | Implant Materials & Artificial Organs |
| Model Number | WZ-MDT-S-01 |
| Instrument classification | Class II |
| Material | Titanium Alloy (Ti6Al4V) |
| Suture Material | UHMWPE |
| Specification | Diameter 1.8x6mm |
| Certificate | CE / ISO |
| Package | Sterile Packaging 1pc/package |
| Application | Orthopedic Surgery / Implantology |

 Tonk Orthopedic Implants
Tonk Orthopedic Implants