




1 / 5










| Still deciding? Get samples of $ ! US$ 100/Piece Request Sample |


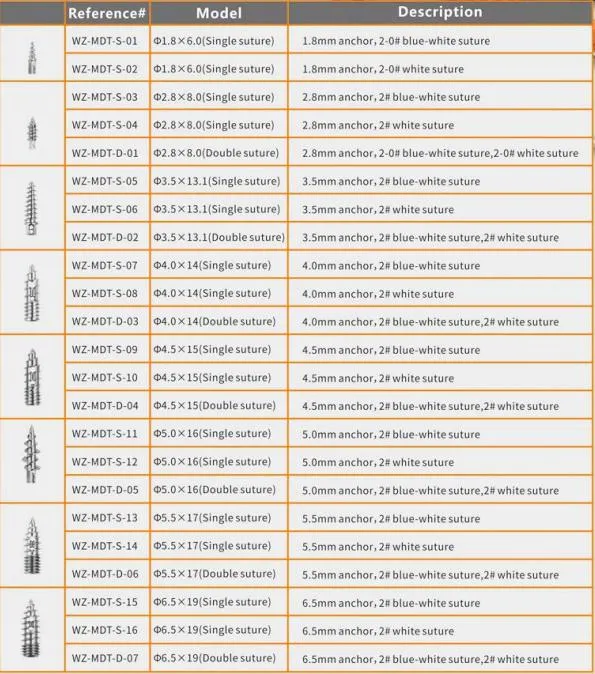
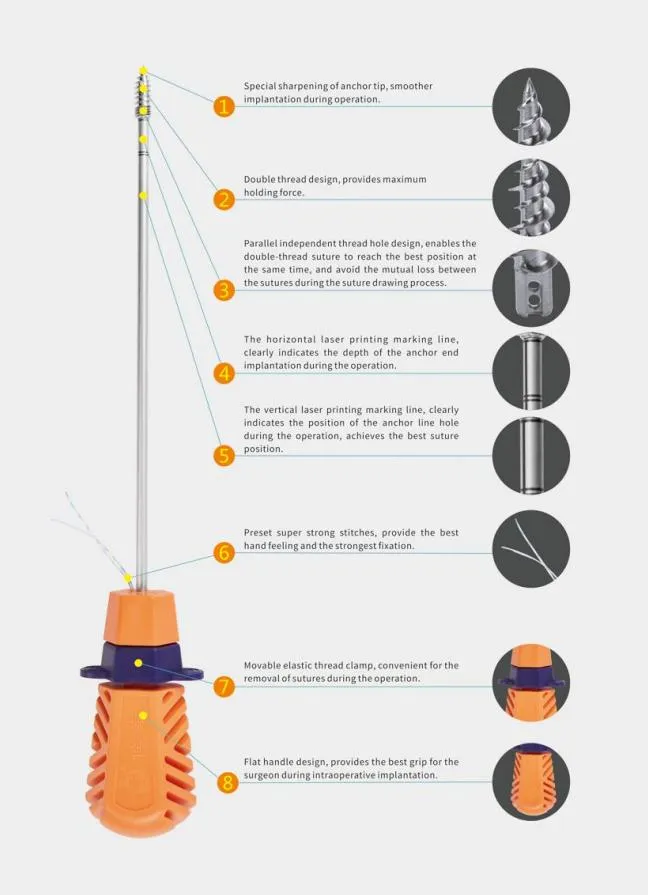
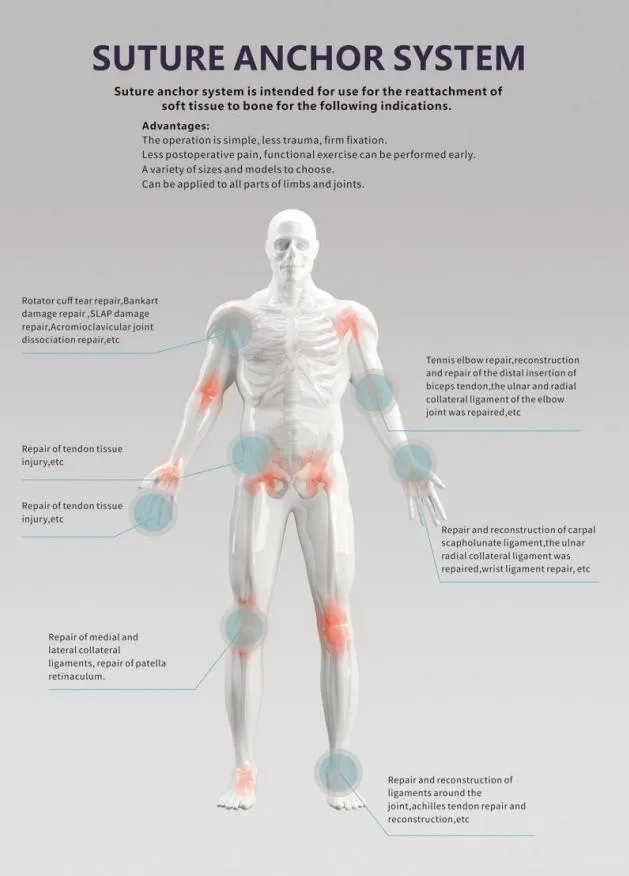
| Product Name | Non-bioabsorbable Suture Anchor |
| Ctn Size | 42*22.5*25.5CM |
| G.W./CTN | 1.5KG-1.8KG |
| Certificates | CE & ISO13485 |
| Validity Period | 3 years |


 Tonk Orthopedic Implants
Tonk Orthopedic Implants