




1 / 5










| Customization: | Available |
|---|---|
| Customized: | Customized |
| Certification: | ISO, CE |




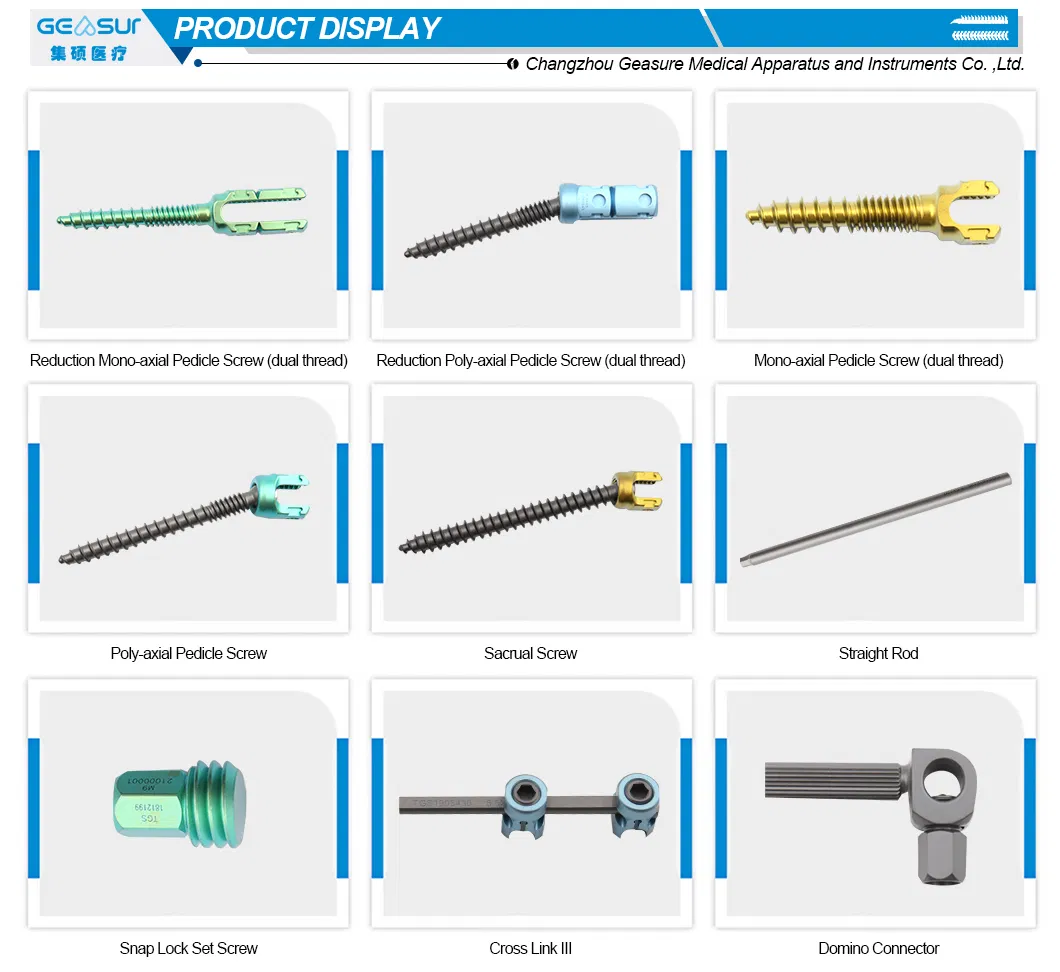
| Posterior Spinal Internal Fixation 5.5 System | |||
| Domino Connector (Material: Titanium) | |||
| Cod (non sterile) | Cod (sterile) | Diameter (mm) | Length (mm) |
| 21005510 | 21085610 | 5.5 | 10 |
| 21005520 | 21085620 | 5.5 | 20 |
| 21005530 | 21085630 | 5.5 | 30 |

Specializing in the research and production of orthopedic implants and instruments, including trauma, spine, joint, and sports medicine. The sales market covers many countries all of the world.
Equipped with first-class production equipments, professional R&D, technology, and manufacturing teams. Established orthopedic laboratory collaborations with university professors and hospital surgeons to provide operational demonstrations.
Obtained ISO13485 system certification and CE certification. Some products have been registered with the FDA 510k. Marketing and sales service strategies are highly recognized by global partners.




 Tonk Orthopedic Implants
Tonk Orthopedic Implants