Product Description
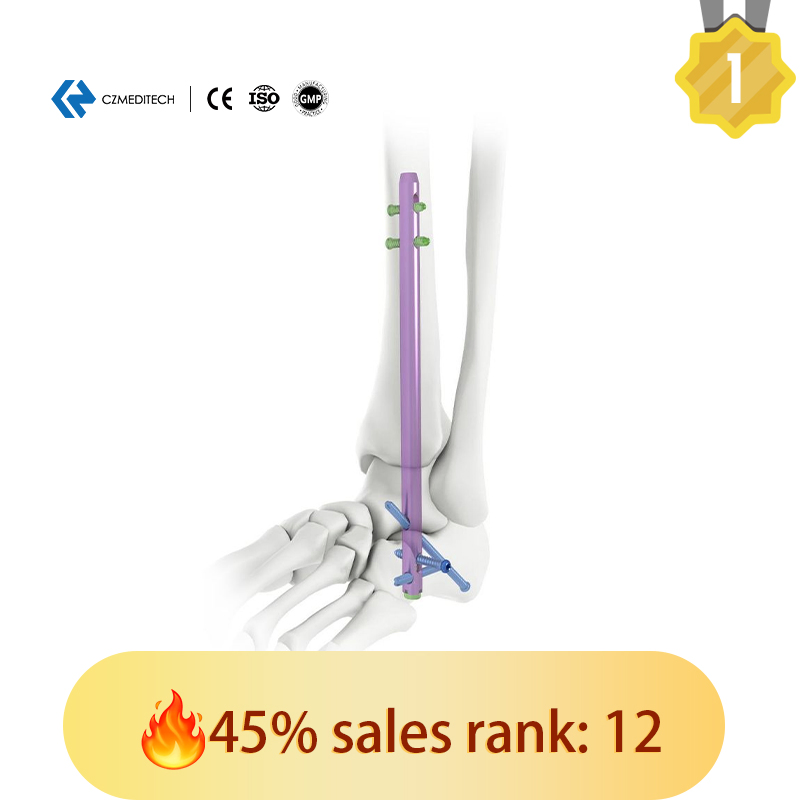
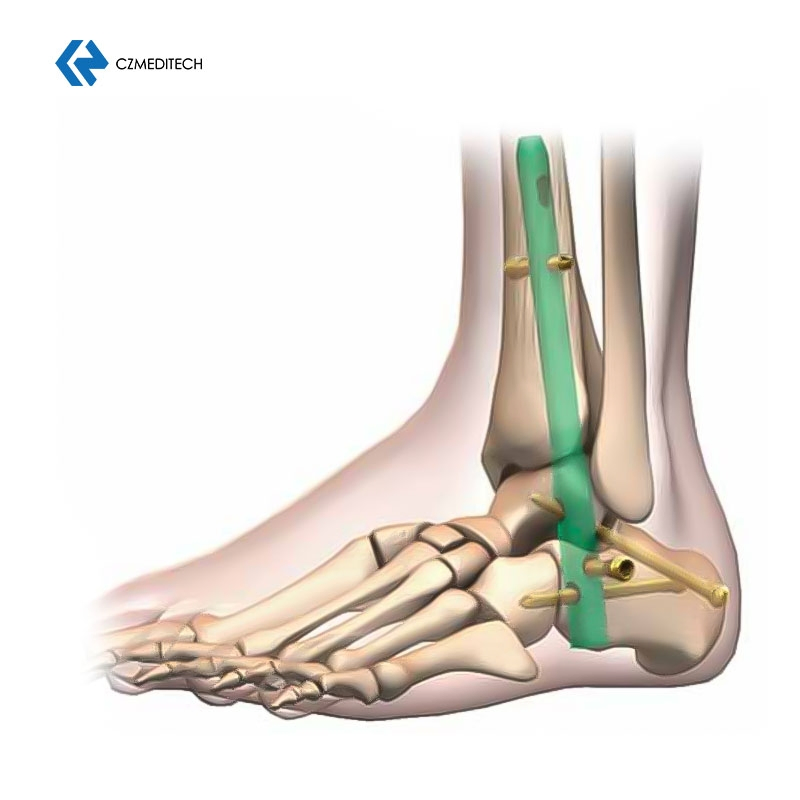
This titanium intramedullary nail is specifically designed for hindfoot and ankle fusion procedures, including tibiotalocalcaneal (TTC) arthrodesis. The interlocking design provides excellent rotational and axial stability, promoting successful fusion. Made from medical-grade titanium alloy (Ti6Al4V), it offers superior biocompatibility, strength, and MRI compatibility.
- Medical-Grade Titanium – Ti6Al4V, biocompatible & MRI compatible
- Interlocking Design – High rotational and axial stability
- Targeted Application – Designed for tibiotalocalcaneal arthrodesis
- Multiple Sizes – Comprehensive diameters and lengths available
- Certified Quality – CE certified meeting international standards
- Customizable – Available for logo engraving and tailored packaging
Product Advantage
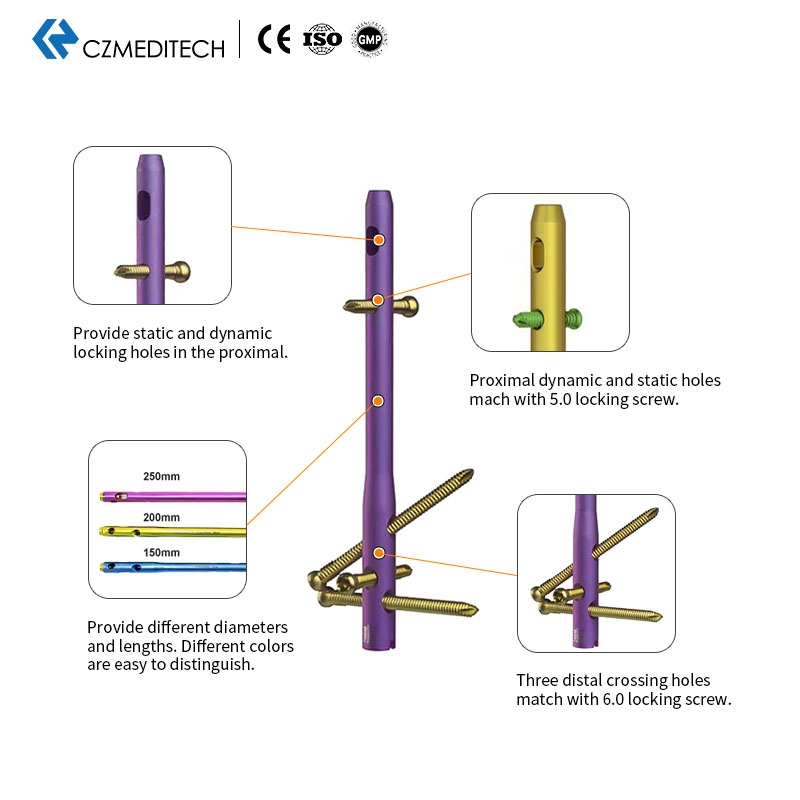
Versatile Range: Various diameters and lengths available with color-coded finishes for easy surgical identification.
Locking Precision: Provides both static and dynamic locking holes in the proximal section.
Screw Compatibility: Proximal holes match 5.0mm locking screws; three distal crossing holes match 6.0mm locking screws.
Professional Capabilities
A Global Leader in Orthopedic Solutions
Successfully supplying to over 2,500 clients in 70+ countries for over 15 years with extensive orthopedic expertise.
One-stop Procurement: Comprehensive portfolio spanning orthopedics, trauma care, and sterilization.
Warranty: 1-year warranty provided with rapid response to all inquiries within 2 days.
CE and ISO 13485 quality management system certified.
Frequently Asked Questions
1. What surgical procedures is this intramedullary nail suitable for?
It is specifically designed for tibiotalocalcaneal (TTC) arthrodesis, ankle fusion, and various hindfoot fusion procedures.
2. What material is used, and is it compatible with MRI scans?
The nail is manufactured from medical-grade titanium alloy (Ti6Al4V), which is lightweight, high-strength, and fully MRI compatible.
3. What size options are available for different patient anatomies?
We provide diameters ranging from 10mm to 12mm and various lengths from 150mm to 250mm. Custom sizes can also be discussed based on specific requirements.
4. Does the nail come with the necessary interlocking screws?
The nail and interlocking screws are typically listed separately. We provide matching 5.0mm and 6.0mm titanium interlocking screws to complete the fixation system.
5. Are the products supplied in sterile packaging?
Yes, we can provide the implants in either sterile (gamma irradiated) or non-sterile formats, depending on your hospital's preference.
6. Do you provide the surgical instrument sets for insertion?
Yes, we offer complete, high-quality instrument sets specifically designed for this nail system, including aiming arms, reamers, and guide wires.






















 Tonk Orthopedic Implants
Tonk Orthopedic Implants