




1 / 5










| Still deciding? Get samples of $ ! US$ 108/Piece Request Sample |
Medical Background: Proximal humeral fractures are among the most common fractures, accounting for 4%–5% of all fractures. Mechanism of injury: Usually occurs when the elbow strikes the ground or the arm is braced during a fall, especially in elderly patients, or when the shoulder joint directly sustains external force.
Interlocking screws with "screw-in-screw" technique; effective for osteoporosis or poor bone quality.
Three screws inserted laterally into the greater tuberosity to ensure basic structural stability.
Ascending screw supports the medial calcar region; two distal screws positioned in different planes.
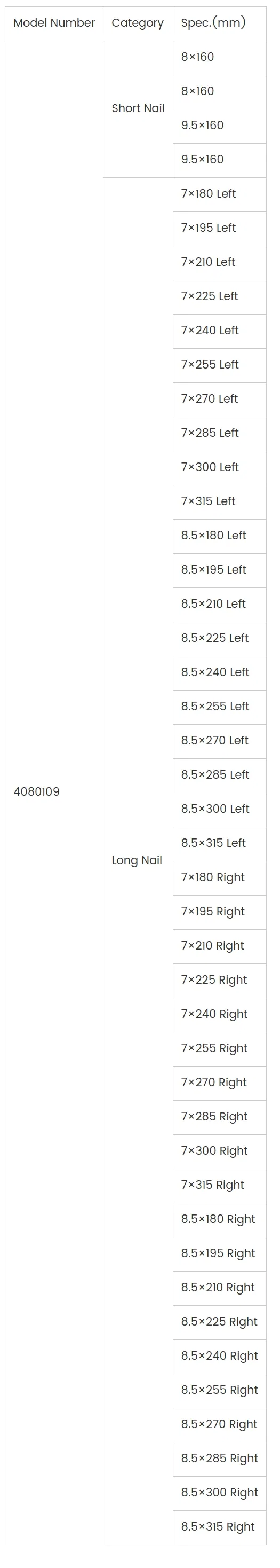
| Short Nail | Length: 160 mm; Diameter: 8 mm, 9.5 mm (L/R). For two-part, three-part, and four-part surgical neck fractures. |
| Long Nail | Length: 180–315 mm; Diameter: 7 mm, 8.5 mm (L/R). For humeral shaft fractures and pathological fractures. |

 Tonk Orthopedic Implants
Tonk Orthopedic Implants