

1 / 3






| Customization: | Available |
|---|---|
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
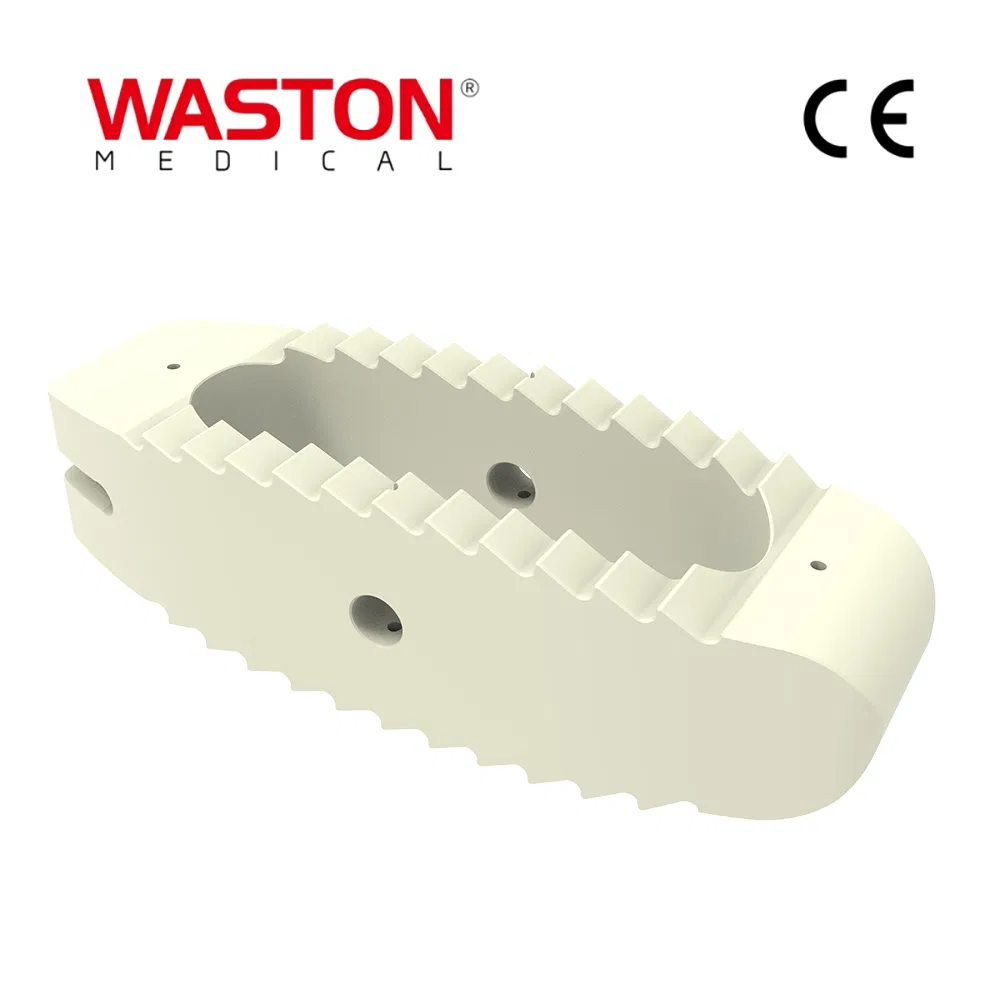
| Model NO. | Cage System |
|---|---|
| Material | Pure Titanium/Titanium Alloy/Stainless Steel |
| Feature | Disposable |
| Certification | CE, ISO |
| Group | Adult |
| Transport Package | Non-Sterile Package, Paper Plastic Roll Bag |
| HS Code | 9021100000 |

| PRODUCT NAME | PRODUCT CODE | DIA | DATA |
|---|---|---|---|
| Laminoplasty Plate | 11101012 | / | H=12 |
| 11101014 | / | H=14 | |
| 11101016 | / | H=16 | |
| Screw (Cross self-tapping) | 10511306 | φ2.5 | L=6 |
| 10511308 | L=8 | ||
| 10511310 | L=10 | ||
| Screw (Cross self-tapping & self-drilling) | 10511406 | φ2.5 | L=6 |
| 10511408 | L=8 | ||
| 10511410 | L=10 |


Operations are strictly aligned with the ISO13485 quality system and medical device manufacturing management specifications, including CE MDD 93/42/EEC directive implementation.
Advanced equipment includes German and American machining centers and Japanese automatic lathes. The Testing Center includes a biomechanical laboratory and physical-chemical testing center for raw material procurement control, product quality control, and new product performance analysis.

Products are exported to many countries including France, Turkey, Indonesia, Mexico, Russia, and Thailand. We also serve as a major OEM supplier to global players in the orthopedic industry.



 Tonk Orthopedic Implants
Tonk Orthopedic Implants