







| Customization: | Available |
|---|---|
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
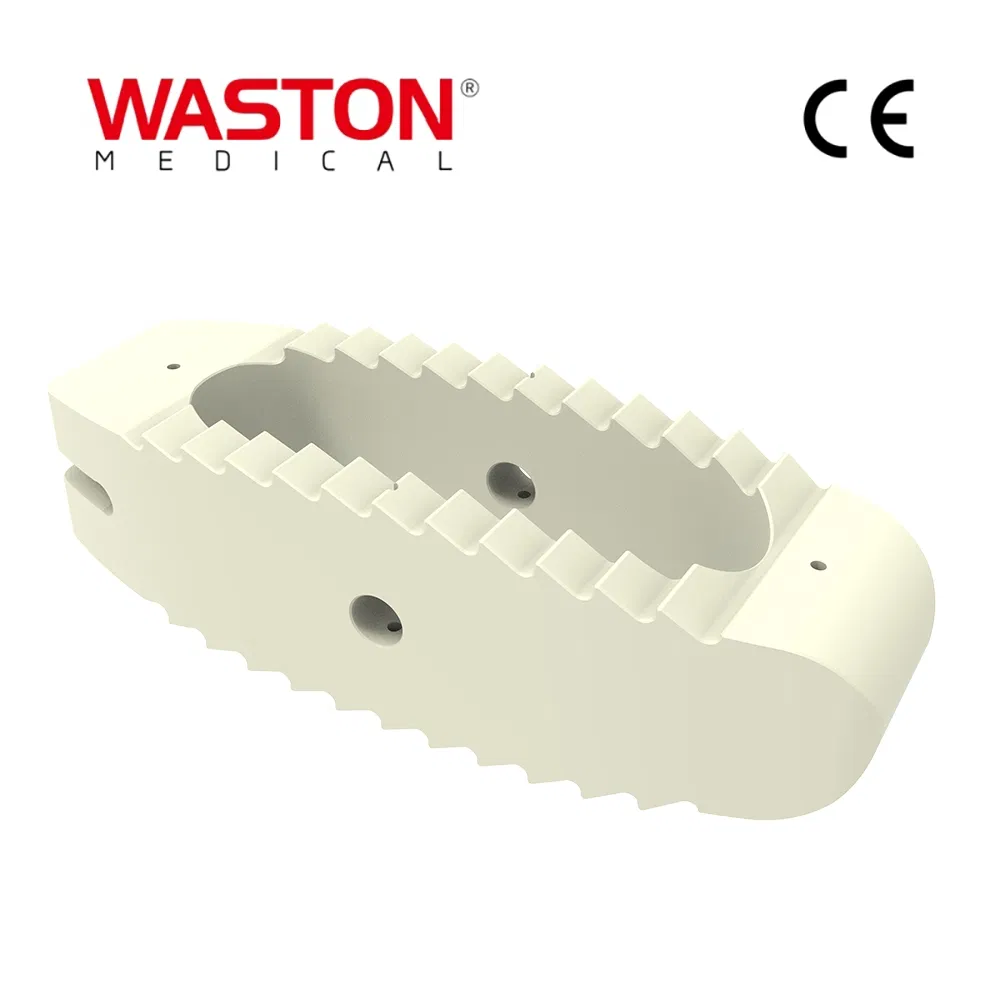
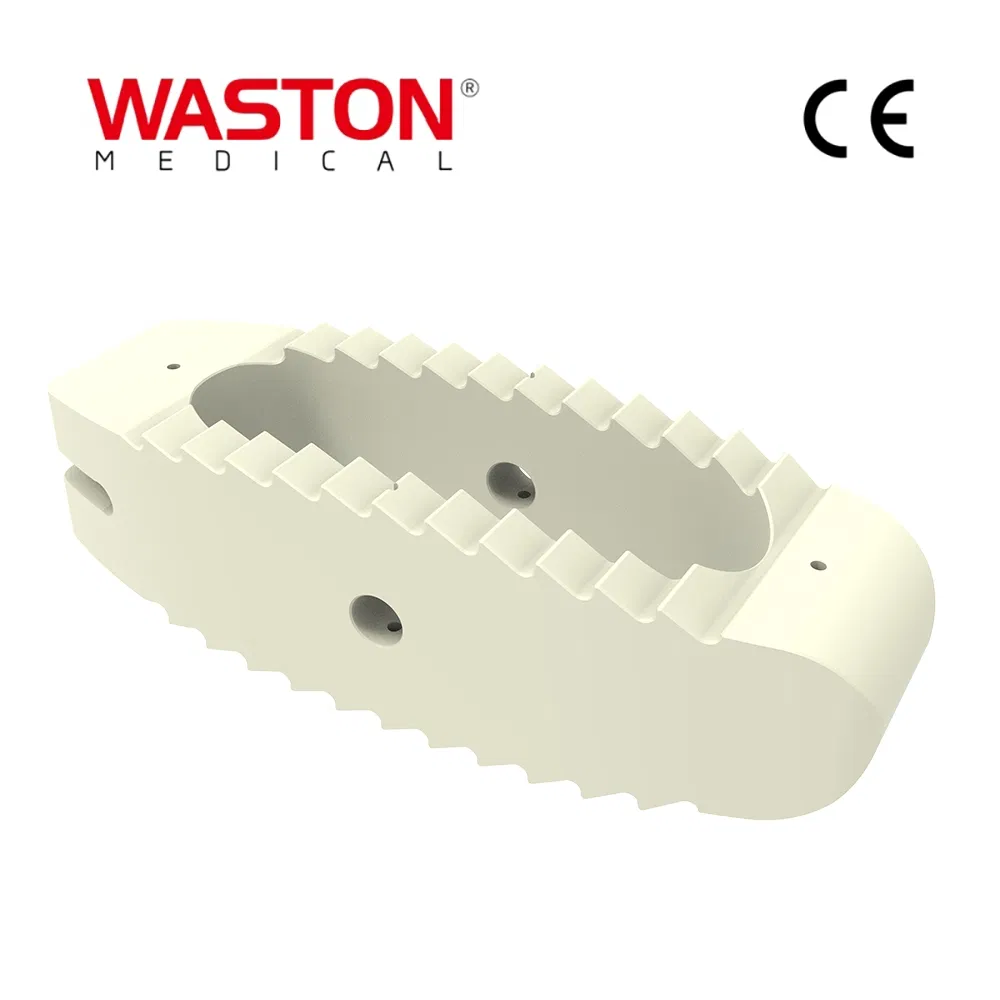
1. Stability: Provide a stable support for vertebral body decompression.
2. Flexibility: Four holes is better for surgery requirement.
3. Design: The unique angle makes vertebral body more stable.
| PRODUCT NAME | PRODUCT CODE | DIA | DATA |
|---|---|---|---|
| NEULEN Laminoplasty Plate | 11101012 | / | H=12 |
| 11101014 | / | H=14 | |
| 11101016 | / | H=16 | |
| NEULEN Screw (Cross self-tapping) | 10511306 | φ2.5 | L=6 |
| 10511308 | L=8 | ||
| 10511310 | L=10 | ||
| NEULEN Screw (Self-tapping & Self-drilling) | 10511406 | φ2.5 | L=6 |
| 10511408 | L=8 | ||
| 10511410 | L=10 |


As a national high-tech enterprise, the proportion of R&D investment to total sales has been over 8% for consecutive years. We pioneer globally revolutionary innovation technologies such as the Rev Drill system, Three-Row-Stapler, Sternal Fixation and Rib plate.

Operating strictly according to the ISO13485 quality system and CE MDD 93/42/EEC directive. We utilize German-imported and American-imported machining centers and Japanese-imported automatic lathes to ensure first-class equipment yields first-class quality.
Our products are exported to many countries, such as Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. We are also a major OEM supplier to some of the largest global players in the orthopaedic industry.



 Tonk Orthopedic Implants
Tonk Orthopedic Implants