




1 / 5










| Still deciding? Get samples of $ ! US$ 258/Set Request Sample |
| Product Name | Model | Part Number | Part Name | Qty |
|---|---|---|---|---|
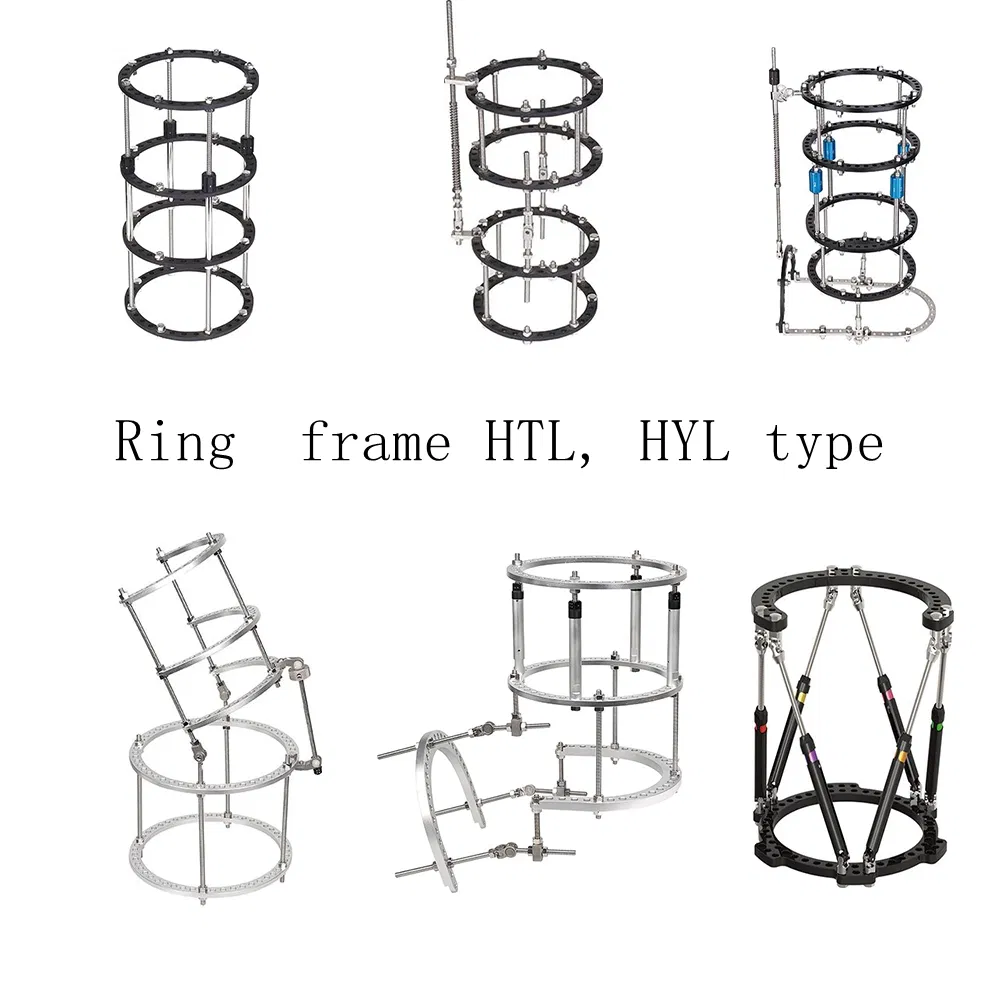
| Lower limb lengthening external fixator | HYL003 | HYLL0108-0126 | Rings | 4 |
| HYLY0108-0112 | Inner threaded connecting rod | 4 | ||
| HYLL05 | Thread rod (M6*150) | 4 | ||
| HYLL05 | Thread rod (M6*120) | 4 | ||
| HYLJ01 | Pin clamp | 16 |



Our organization is a long-standing leader in the development, manufacture, and sales of orthopedic implants and instruments. As a key enterprise in the medical apparatus industry, we offer a comprehensive product line that includes spinal pedicle screws, interlocking nails, locking plates, trauma screws, external fixators, and surgical instruments.
Equipped with advanced imported CNC machinery and high-precision digital control systems, our production facility maintains a 100,000 Grade purification chamber to ensure the highest product quality. We are committed to "perfect design, innovative technology, and top-ranking products," backed by a robust quality management system certified under ISO 9001 and CE standards.



 Tonk Orthopedic Implants
Tonk Orthopedic Implants