


1 / 5










| Customization: | Available |
|---|---|
| Certification: | FDA, ISO, CE |
| Disinfection: | Without Disinfection |

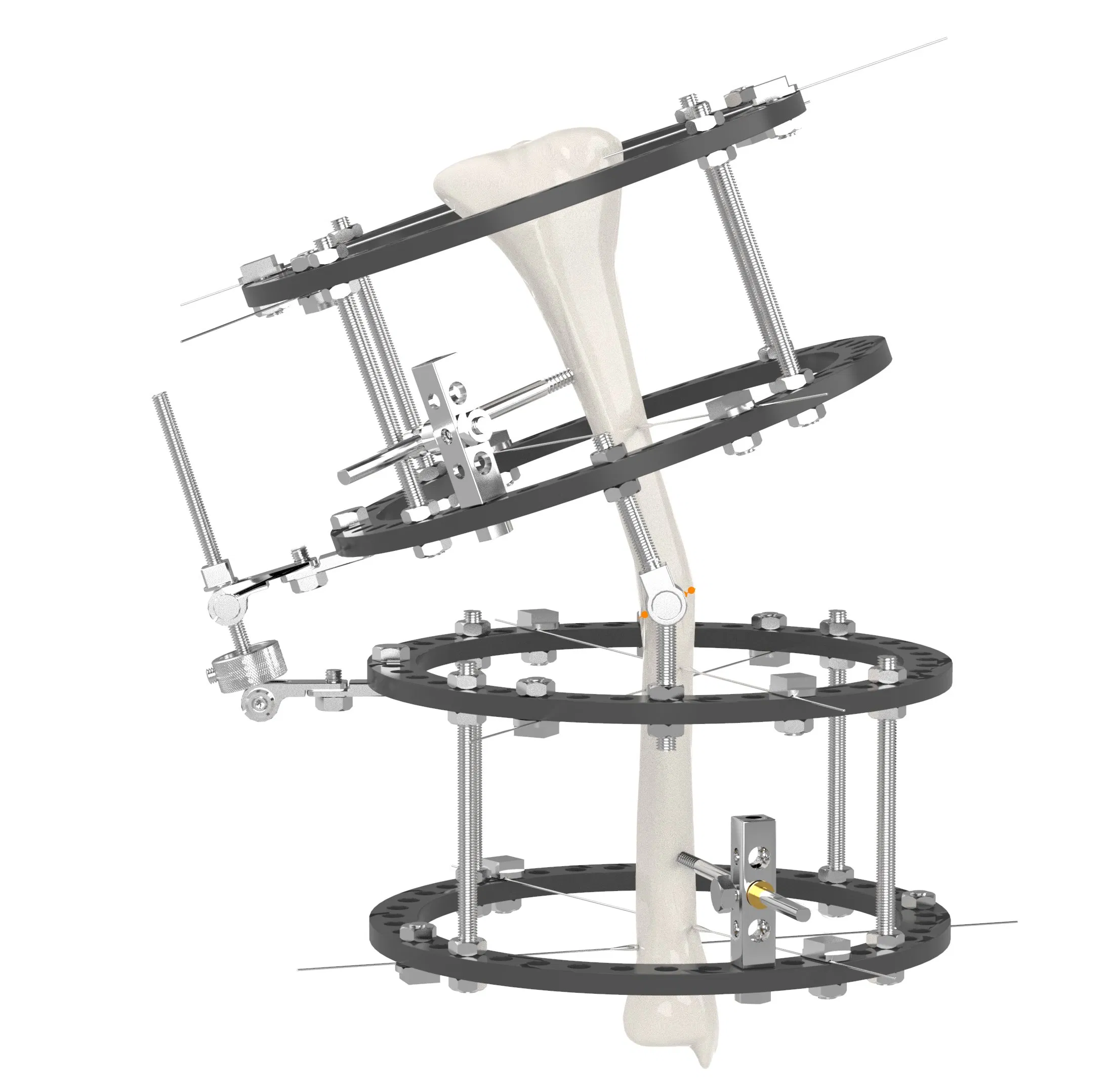
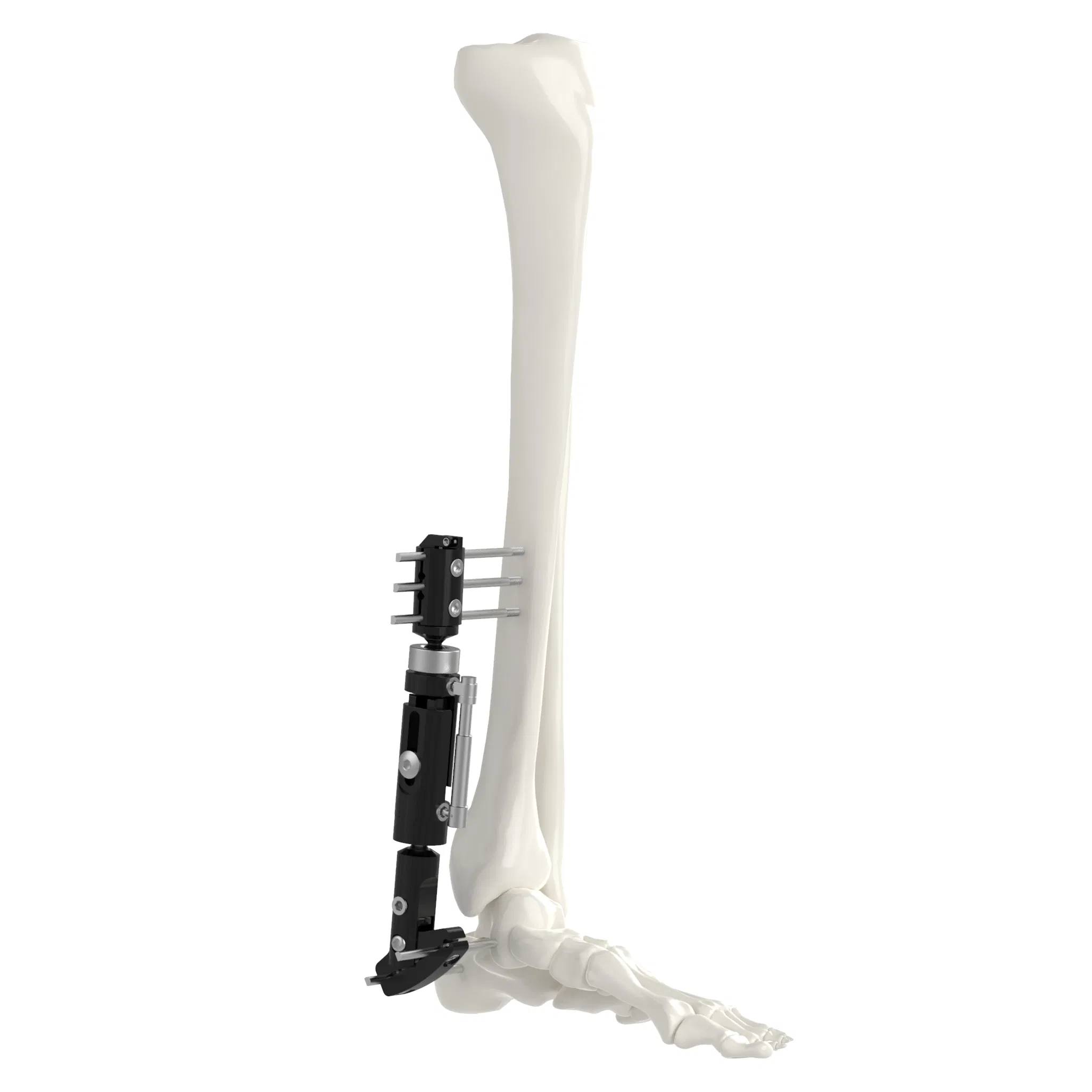
| System Overview | |||
| Single arm | Combine | Ilizarov | Needle |




Founded in 1994, this medical device enterprise has grown to become one of the largest designers, manufacturers, and suppliers of orthopedic implants and instruments in the region. The facility encompasses over 60,000 square meters of modern workshop space.
Specializing in traumatology, spinal fixation systems, and surgical instruments, the product portfolio includes femoral nails, titanium plates, screws, pedicle screws with rods, PEEK cages, and balloon kyphoplasty systems. These high-quality medical solutions are distributed globally across Europe, Latin America, the Middle East, and Asia.

 Tonk Orthopedic Implants
Tonk Orthopedic Implants