 5.5/6.5 Orthopedic Series Spinal System Pedicle Spinal Screws
5.5/6.5 Orthopedic Series Spinal System Pedicle Spinal Screws
 Orthopedic Titanium Pedicle Screw Instrument Set 6.0mm
Orthopedic Titanium Pedicle Screw Instrument Set 6.0mm
 Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw Set
Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw Set
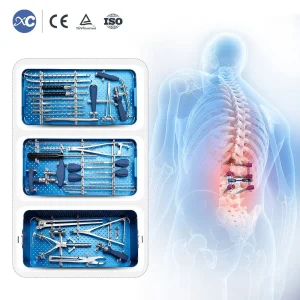 Orthopedic Bone Surgical Set 6.0mm Spinal Pedicle System
Orthopedic Bone Surgical Set 6.0mm Spinal Pedicle System
 Dial-Adjustable Weight Stack System with Chrome Plated Guide Rods
Dial-Adjustable Weight Stack System with Chrome Plated Guide Rods
 Dial-Adjustable Weight Stack System with Chrome Guide Rods
Dial-Adjustable Weight Stack System with Chrome Guide Rods
 Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm
Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm
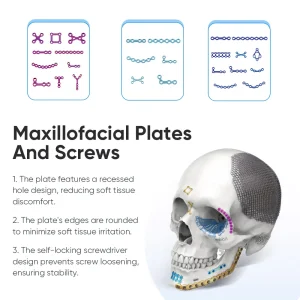 Orthopedic Titanium Maxillofacial Internalfixation System 1.5mm/2.0mm
Orthopedic Titanium Maxillofacial Internalfixation System 1.5mm/2.0mm
The global orthopedic market has witnessed a paradigm shift toward advanced biomaterials, with Titanium Spinal Rods standing at the forefront of spinal fusion and deformity correction. As the world population ages, the prevalence of degenerative disc diseases, spinal stenosis, and osteoporotic fractures has skyrocketed, fueling an unprecedented demand for reliable, biocompatible, and high-strength fixation systems.
Currently, the market is valued in the billions, with North America and Europe leading in consumption, while the Asia-Pacific region emerges as the fastest-growing hub for surgical procedures. Titanium (specifically Grade 5 and Grade 23 ELI) remains the "gold standard" due to its exceptional strength-to-weight ratio, resistance to corrosion, and MRI compatibility—critical factors for long-term patient recovery and monitoring.
Annual Market Growth
Medical Grade Standard
The integration of 3D printing allows for the creation of porous titanium structures that mimic human bone architecture. This encourages "Osseointegration," where natural bone grows directly into the implant, providing superior stability compared to traditional smooth rods.
Modern surgery is moving toward patient-specific rods. By utilizing AI and pre-operative CT scans, manufacturers can now pre-bend titanium rods to the exact curvature of a patient’s unique anatomy, reducing surgical time and improving clinical outcomes.
Emerging trends include coating titanium rods with Hydroxyapatite (HA) or antimicrobial agents to reduce the risk of post-operative infections and accelerate the healing process in complex spinal reconstructions.
Procurement officers in hospitals and medical distribution firms prioritize CE, FDA, and ISO 13485 certifications. As a Chinese manufacturer, meeting these stringent international standards is non-negotiable for entering the global tier-1 medical supply chain.
In a post-pandemic world, buyers are looking for "One-Stop" suppliers. They require a partner who can provide not just the titanium rods, but also the pedicle screws, connectors, and the specialized instrument sets required for implantation.
With healthcare budgets tightening globally, there is a massive shift toward high-quality "Value-Based" implants. Manufacturers who can offer Tier-1 quality at Tier-2 pricing are winning significant tenders in emerging markets like Latin America, SE Asia, and the Middle East.
Our Titanium Spinal Rods are engineered for versatility across various surgical environments and regional needs.
In high-volume emergency hospitals, the speed of fixation is vital. Our 6.0mm systems with intuitive instrument sets allow surgeons to stabilize spinal fractures quickly and effectively.
Correction of adolescent idiopathic scoliosis requires rods that provide high corrective force while maintaining fatigue resistance. Our high-strength titanium alloys offer the perfect balance for growing spines.
For elderly patients with osteoporotic bone, we provide cannulated cement-augmented screw systems compatible with our titanium rods to ensure fixation in compromised bone structures.
Choosing a China Wholesale Titanium Spinal Rods Manufacturer offers distinct strategic advantages for global distributors:
OMA was established in the year 1981 and has since been engaged in the export of Medical equipments, Hospital equipments, Orthopaedic Implants & instruments, Laboratory equipments, Scientific & Educational products worldwide.
Our goods have reached all continents from "The Americas" to "Europe" and beyond…
With 100+ employees who are involved in activities related to production, inspections and dispatch we assure you world class healthcare products delivered to your doorstep. Each employee has in-depth knowledge about the product and has years of experience to back that knowledge.
The product range includes Orthopaedic Implants, Hospital Hollow wares, Resuscitators, Laryngoscopes, Hospital Furniture, Rehabilitation Aids, Autoclaves, Sterilizers, Suction Machines and many more.
We offer you a complete experience starting from the placement of your order until the successful installation of the product. The logistics department makes sure that your goods reach you with the best available carriers in the shortest possible time frame. Our post-sales customer service team is available 24/7 to answer all your queries regarding our products.


The logistics department makes sure that your goods reach you with the best available carriers in the shortest possible time frame.

Every OMA product comes with an assurance of high quality and zero compromise in its production, performance, and safety.

Our innovative approach towards product design and production help us bring you the best products at even better prices.

Our expert post-sales customer service team is available 24/7 to promptly answer all your queries regarding our products.
 Orthopedic Spinal Fixation Medical Instrument Vertebral Hook
Orthopedic Spinal Fixation Medical Instrument Vertebral Hook
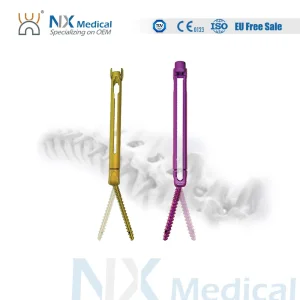 Nx Medical Orthopedic Implant Cannulated Bone Cement Spinal Pedicle Screw
Nx Medical Orthopedic Implant Cannulated Bone Cement Spinal Pedicle Screw
 Posterior Thoracolumbar Spinal Fixation System Titanium Surgery
Posterior Thoracolumbar Spinal Fixation System Titanium Surgery
 Orthopedic Spinal Fixation Medical Instrument Spine Vertebral Hook
Orthopedic Spinal Fixation Medical Instrument Spine Vertebral Hook
 Orthopedic Spinal Fixation Vertebral Hook Medical Instrument
Orthopedic Spinal Fixation Vertebral Hook Medical Instrument
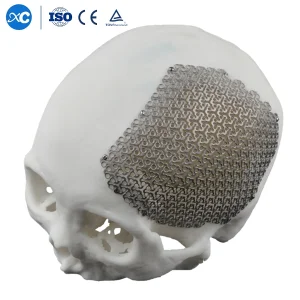 3D Anatomical Cloud Shape Cmf Cranial Mesh Titanium Implant
3D Anatomical Cloud Shape Cmf Cranial Mesh Titanium Implant
 Master 10 Spinal System, Titanium Alloy, Orthopedic Implant Set
Master 10 Spinal System, Titanium Alloy, Orthopedic Implant Set
 Master 10 Spinal System Titanium Alloy Orthopedic Implant Spine Surgical
Master 10 Spinal System Titanium Alloy Orthopedic Implant Spine Surgical
When searching for a China Wholesale Titanium Spinal Rods Manufacturer, technical specifications are the most critical variable. Our spinal rods are primarily manufactured from Ti-6Al-4V ELI (Extra Low Interstitial), often referred to as Grade 23. This material is superior to standard Grade 5 titanium because it offers higher ductility and better fracture toughness, which is essential when a surgeon is contouring a rod to fit the patient's spinal anatomy during a posterior fixation procedure.
The manufacturing process involves rigorous stages of quality control. It begins with raw material verification through spectral analysis to ensure the chemical composition meets ASTM F136 standards. Following this, the rods undergo precision centerless grinding to achieve a perfect diameter (typically 5.5mm or 6.0mm) with a tolerance within microns. This precision ensures that the rods interface perfectly with pedicle screws and cross-links, preventing "notching" or mechanical failure under the cyclic loads of daily human movement.
Furthermore, our surface treatment technologies include electro-polishing and anodization. Anodization not only provides color-coding (useful for differentiating rod diameters in the OR) but also increases the thickness of the natural titanium oxide layer, further enhancing its already legendary biocompatibility. This ensures that the implant remains inert within the body for decades, minimizing the risk of "metallosis" or adverse tissue reactions.
From a clinical perspective, the stiffness of the rod is a double-edged sword. While rigidity is required for stabilization, a rod that is too stiff can cause "stress shielding," where the implant carries all the load and the surrounding bone weakens. Our engineering team has optimized the modulus of elasticity in our titanium rods to closely approximate the physiological properties of human cortical bone, promoting healthy load-sharing and faster fusion rates.
In complex cases such as adult spinal deformity (ASD) or neuromuscular scoliosis, our rods are used in tandem with multi-axial pedicle screws and lateral connectors. The "Master 10" and "Orthopedic 6.0mm" series mentioned above are designed to withstand the massive corrective forces required to bring a spine back into sagittal balance. By choosing a manufacturer that understands these biomechanical nuances, hospital procurement teams can ensure they are providing surgeons with tools that translate directly into higher patient satisfaction scores and lower revision surgery rates.
The next decade of spinal surgery will be defined by the "Digital Operating Room." We are currently investing in smart manufacturing technologies that will allow for digital tracking of every single spinal rod from the forge to the operating table. Through QR-coded laser marking, hospitals can instantly access the material certificates and sterilization history of each implant, ensuring total transparency and safety.
As a leading supplier, we are also exploring the development of "Cobalt-Chrome" (CoCr) rods for specific high-correction maneuvers where even higher stiffness than titanium is required. This expansion of our product portfolio ensures that our global partners can source every possible configuration for spinal health from a single, trusted Chinese factory.